|
|
|
|
 |
RODENTS: Voles |
|
|

Identification
Voles, also called meadow
mice or field mice, belong to the genus Microtus. Voles
are compact rodents with stocky bodies, short legs, and
short tails. Their eyes are small and their ears
partially hidden. Their underfur is generally dense and
covered with thicker, longer guard hairs. They usually
are brown or gray, though many color variations exist.
There are 23 vole species
in the United States. This chapter provides range maps,
descriptions, and habitat characteristics for seven
species that are widespread or cause significant
economic damage. Tentative identification of a
particular animal may be made using this information.
For positive identification, use a field guide or
contact an expert.
Prairie Vole (Microtus
ochrogaster). The prairie vole is 5 to 7 inches (13
to 18 cm) in total length (nose to tip of tail). Its fur
is gray to dark brown and mixed with gray, yellow, or
hazel-tipped hairs, giving it a “peppery” appearance.
Underparts are gray to yellow-gray. It is the most
common vole in prairie habitats.
Meadow Vole (M.
pennsylvanicus). The meadow vole is the most widely
distributed Microtus species in the United States. Its
total length is 5 1/2 to 7 1/2 inches (14 to 19 cm) and
its fur is gray to yellow-brown, obscured by
black-tipped hairs. Northern subspecies may also have
some red in their fur. Its underparts are gray, at times
washed with silver or buff. The tail is bicolored.
Long-tailed Vole (M.
longicaudus). The long-tailed vole can be
distinguished from other Microtus species by its tail,
which comprises 30% or more of its total length of 6 to
8 1/2 inches (15 to 21 cm). The long-tailed vole has
gray to dark brown fur with many black-tipped hairs. The
underparts are gray mixed with some white or yellow. The
tail is indistinctly to sharply bicolored.
Pine or Woodland Vole (M.
pinetorum). The pine vole is a small vole. Its total
length is 4 to 6 inches (10 to 15 cm). Its brown fur is
soft and dense. The underparts are gray mixed with some
yellow to cinnamon. The tail is barely bicolored or
unicolored.
Montane (or Mountain) Vole
(M. montanus). The montane vole is 5 1/2 to 8 1/2 inches
(15 to 20 cm) in total length. Its fur is brown, washed
with gray or yellow, and mixed with some black-tipped
hairs. Its feet are usually silver-gray and its body
underparts are whitish. The tail is bicolored.
Oregon Vole (M. oregoni).
The Oregon vole is 5 1/2 to 6 1/2 inches (14 to 16 cm)
in length. Its fur is gray to brown or yellow-brown.
Underparts are darkish, washed with yellow to white. The
tail is indistinctly bicolored.
California Vole (M.
californicus). The California vole is 6 to 8 1/2 inches
(15 to 20 cm) in total length. Its fur is tawny olive to
cinnamon brown with brown to black overhairs. The
underparts are grayish. The tail is bicolored.
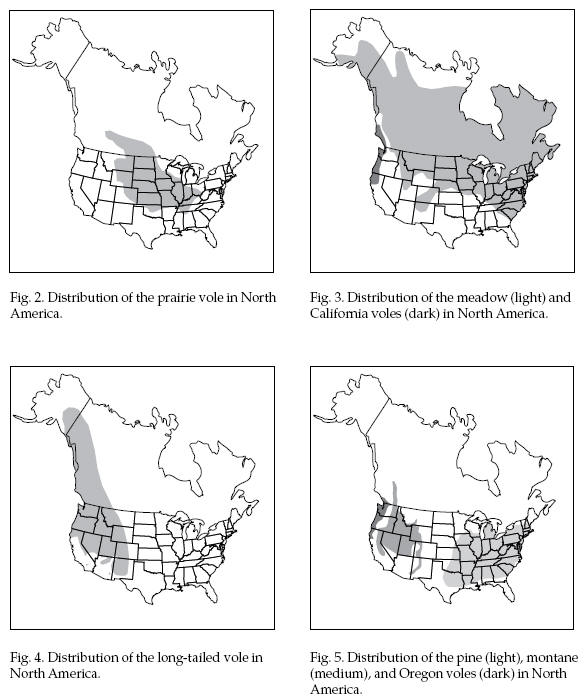
Range
Figures 2, 3, 4, and 5
show the approximate ranges of these species.
Habitat
Voles occupy a wide
variety of habitats. They prefer areas with heavy ground
cover of grasses, grasslike plants, or litter. When two
species are found together in an area, they usually
occupy different habitats. Though voles evolved in
“natural” habitats, they also use habitats modified by
humans, such as orchards, windbreaks, and cultivated
fields, especially when vole populations are high.
Characteristic habitat descriptions for the seven
described species follow.
Prairie Vole. The prairie
vole, as the name suggests, is the most common vole of
the Great Plains grasslands. It is found in a variety of
habitats, such as old fields, marshlands, and grass
prairies. When in association with the meadow vole, it
is generally in drier habitats.
Meadow Vole. The meadow
vole is found in the northern United States and Canada.
It prefers wet meadows and grassland habitats. When in
association with the montane vole or prairie vole, it is
generally in moister habitats.
Long-tailed Vole. The
long-tailed vole is found in a wide variety of habitats
(for example, sagebrush grasslands, forests, mountain
meadows, and stream banks) in the western United States
and Canada.
Pine Vole. The pine vole
is found in the eastern United States. It inhabits a
variety of habitats such as deciduous and pine forests,
abandoned fields, and orchards. Heavy ground cover is
characteristic of these habitats.
Montane Vole. The montane
vole is found primarily in mountainous regions of the
western United States. It is found in alpine meadows,
dry grasslands, and sagebrush grasslands. It avoids
forests. When in association with the meadow vole, it is
generally in drier habitats.
Oregon Vole. The Oregon
vole is most often found in forested areas of northern
California, Oregon, and Washington where there is an
understory of forbs and grasses such as in burned or
clear-cut areas.
California Vole. The
California vole inhabits the chaparral woodland
shrubland of California. It is found in both wet and
well-drained areas.
Food Habits
Voles eat a wide variety
of plants, most frequently grasses and forbs. In late
summer and fall, they store seeds, tubers, bulbs, and
rhizomes. They eat bark at times, primarily in fall and
winter, and will eat crops, especially when their
populations are high. Occasional food items include
snails, insects, and animal remains.
General Biology, Reproduction, and Behavior
Voles are active day and
night, year-round. They do not hibernate. Home range is
usually 1/4 acre (0.1 ha) or less but varies with
season, population density, habitat, food supply, and
other factors. Voles are semifossorial and construct
many tunnels and surface runways with numerous burrow
entrances. A single burrow system may contain several
adults and young.
Voles may breed throughout
the year, but most commonly in spring and summer. In the
field, they have 1 to 5 litters per year. They have
produced up to 17 litters per year in a laboratory.
Litter sizes range from 1 to 11, but usually average 3
to 6. The gestation period is about 21 days. Young are
weaned by the time they are 21 days old, and females
mature in 35 to 40 days. Lifespans are short, probably
ranging from 2 to 16 months. In one population, there
was 88% mortality during the first month of life.
Large population
fluctuations are characteristic of voles. Population
levels generally peak every 2 to 5 years; however, these
cycles are not predictable. Occasionally during
population irruptions, extremely high vole densities are
reached. Dispersal, food quality, climate, predation,
physiological stress, and genetics have been shown to
influence population levels. Other factors probably also
play a part.
Population densities are
variable. Smolen and Keller (1987) list densities of
long-tailed vole populations. A California population
ranged from about 2 to 7 voles per acre (5 to 16/ha) and
a New Mexico population ranged from around 8 to 49 voles
per acre (20 to 121/ha). Cole and Batzli (1979) found
that prairie vole populations averaged 15 per acre
(38/ha) in prairie, 52 per acre (128/ha) in bluegrass,
and 99 per acre (244/ha) in alfalfa. Another vole
population ranged from 1 to 14 per acre (2 to 35/ha)
over 3 years in western mixed prairie. Variability in
meadow vole population density was reported by Taitt and
Krebs (1985). An Ontario, Canada population ranged from
32 to 162 per acre (80 to 400/ha) over 1 year while an
Illinois population ranged from 2 to 6 per acre (5 to
15/ha) also over 1 year. Other populations show similar
year-to-year variability. Much higher densities may be
reached during population irruptions. In Klamath Basin,
Oregon, montane vole densities ranged from 200 to 500
per acre (500 to 1,250/ha) and may have reached 4,000
per acre (10,000/ha) in some instances during a 1957 to
1958 irruption.
Many voles are excellent
swimmers. The water vole, in fact, escapes predators by
swimming and diving. The climbing ability of voles
varies. The long-tailed vole, for example, is a good
climber (Johnson and Johnson 1982) while the pine vole
is a bit clumsy in this regard.
Voles are prey for many
predators (for example, coyotes, snakes, hawks, owls,
and weasels); however, predators do not normally control
vole populations.
Damage and Damage Identification
Voles may cause extensive
damage to orchards, ornamentals, and tree plantings due
to their girdling of seedlings and mature trees.
Girdling damage usually occurs in fall and winter. Field
crops (for example, alfalfa, clover, grain, potatoes,
and sugar beets) may be damaged or completely destroyed
by voles. Voles eat crops and also damage them when they
build extensive runway and tunnel systems. These systems
interfere with crop irrigation by displacing water and
causing levees and checks to wash out. Voles also can
ruin lawns, golf courses, and ground covers.
Girdling and gnaw marks
alone are not necessarily indicative of the presence of
voles, since other animals, such as rabbits, may cause
similar damage. Vole girdling can be differentiated from
girdling by other animals by the non-uniform gnaw marks.
They occur at various angles and in irregular patches.
Marks are about 1/8 inch (0.3 cm) wide, 3/8 inch (1.0
cm) long, and 1/16 inch (0.2 cm) or more deep. Rabbit
gnaw marks are larger and not distinct. Rabbits neatly
clip branches with oblique clean cuts. Examine girdling
damage and accompanying signs (feces, tracks, and burrow
systems) to identify the animal causing the damage.
The most easily
identifiable sign of voles is an extensive surface
runway system with numerous burrow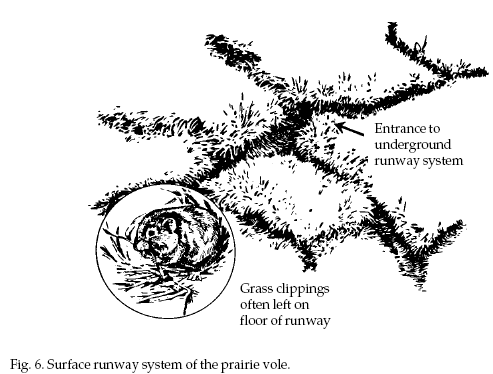 opening (Fig. 6). Runways are 1 to 2 inches (2.5 to 5
cm) in width. Vegetation near well-traveled runways may
be clipped close to the ground. Feces and small pieces
of vegetation are found in the runways.
opening (Fig. 6). Runways are 1 to 2 inches (2.5 to 5
cm) in width. Vegetation near well-traveled runways may
be clipped close to the ground. Feces and small pieces
of vegetation are found in the runways.
The pine vole does not use
surface runways. It builds an extensive system of
underground tunnels. The surface runways of long-tailed
voles are not as extensive as those of most other voles.
Voles pose no major public
health hazard because of their infrequent contact with
humans; however, they are capable of carrying disease
organisms, such as plague (Yersinia pestis) and
tularemia (Francisilla tularensis). Be careful and use
protective clothing when handling voles.
Legal Status
Voles are classified as
nongame mammals and can be controlled when causing
damage. Contact your local state wildlife agency for
details regarding applicable codes and regulations.
Damage
Prevention and Control Methods
Exclusion
Hardware cloth cylinders
exclude voles from seedlings and young trees. The mesh
should be 1/4 inch (0.6 cm) or less in size. Bury the
wire 6 inches (15 cm) to keep voles from burrowing under
the cylinder. Large scale fencing of areas is probably
not cost-effective. Drift fences with pit traps may be
used to monitor populations and can indicate when voles
are immigrating to crops, orchards, or other cultivated
areas.
Cultural Methods and
Habitat Modification
Cultural and habitat
modification practices can reduce the likelihood and
severity of vole damage. Eliminate weeds, ground cover,
and litter in and around crops, lawns, and cultivated
areas to reduce the capacity of these areas to support
voles. Lawn and turf should be mowed regularly. Mulch
should be cleared 3 feet (1 m) or more from the bases of
trees.
Voles can live in dense
populations in ditch banks, rights-of-way, and water
ways that are unmanaged. Adjacent crop fields can be
cost-effectively protected by controlling vegetation
through mowing, spraying, or grazing.
Soil tillage is effective
in reducing vole damage as it removes cover, destroys
existing runway-burrow systems and kills some voles
outright. Because of tillage, annual crops tend to have
lower vole population levels than perennial crops. Voles
are nevertheless capable of invading and damaging annual
crops, especially those that provide them with cover for
extended periods of time.
Frightening
Frightening agents are not
effective in reducing vole damage.
Repellents
Repellents utilizing
thiram (also a fungicide) or capsaicin (the “hot” in
chilis) as an active ingredient are registered for
meadow voles (see Supplies and Materials). These
products (or repellents registered for other species)
may afford short-term protection, but this has not been
demonstrated. Check with your state pesticide regulatory
agency for availability.
Toxicants
Zinc phosphide is the most
commonly used toxicant for vole control. It is a
single-dose toxicant available in pelleted and grain
bait formulations and as a concentrate. Zinc phosphide
baits generally are broadcast at rates of 6 to 10 pounds
per acre (7 to 11 kg/ ha), or are placed by hand in
runways and burrow openings. Although prebaiting
(application of similar nontreated bait prior to
applying toxic bait) is not usually needed to obtain
good control, it may be required in some situations,
such as when a population has been baited several times
and bait shyness has developed. Zinc phosphide baits are
potentially hazardous to ground-feeding birds,
especially waterfowl. Placing bait into burrow openings
may reduce this hazard.
Anticoagulant baits are
also effective in controlling voles. Anticoagulants are
slow-acting toxicants requiring from 5 to 15 days to
take effect. Multiple feedings are needed for most
anticoagulants to be effective. In many states, one or
more anticoagulant baits are registered for controlling
voles.
In addition to broadcast
and hand placement, anticoagulant baits also can be
placed in various types of bait containers (Byers and
Merson 1982, Radvanyi 1980). Water repellent paper tubes
with an anticoagulant bait glued to the inside surface
make effective, disposable bait containers. Tube size is
about 5 inches (12 cm) long by 1 1/2 inches (4 cm) in
diameter (Libby and Abrams 1966, Marsh et al. 1967).
Bait containers protect bait from moisture and reduce
the likelihood of nontarget animals and small children
consuming bait.
Fumigants
Fumigants usually are not
effective because the complexity and shallowness of vole
burrow systems allow the fumigant to escape. They may
work in new, small burrow systems with only one or two
entrances.
Trapping
Trapping is not effective
in controlling large vole populations because time and
labor costs are prohibitive. Mouse snap traps can be
used to control a small population by placing the trap
perpendicular to the runway with the trigger end in the
runway. A peanut butter-oatmeal mixture or apple slices
make good baits. Fall and late winter are periods when
many vole species are easiest to trap.
Although voles rarely
invade houses, in the event that they do, they can be
controlled by setting snap traps or live traps (Sherman
or box-type) as you would for house mice (see Trapping
in the House Mice chapter).
Shooting
Shooting is not practical
or effective in controlling voles.
Other Methods
A wide variety of
predators feed on voles. Voles are relatively easy for
most predators to catch and are active, and therefore
available, day and night year-round. Despite their
vulnerability and availability, voles are not usually
“controlled” by predators. This is because voles have a
high reproductive potential. Postpartum breeding is
common and females may breed as early as 2 weeks of age.
Synchronous breeding also occurs. These factors enable
voles to increase at a faster rate than predators
(Pearson 1985).
Economics of Damage and Control
Jameson (1958) calculated
that 100 meadow voles per acre destroyed about 4% of an
alfalfa crop, which amounted to about 1,000 pounds per
acre (1,136 kg/ha) over 7 months.
Populations of 1,700 voles
per acre (4,250 voles/ha) in Washington State apple
orchards decreased production by 35%. This amounted to a
loss of $3,036 per acre ($7,590/ha) due to reduced fruit
quality and quantity. One year after eliminating voles,
the production in the orchard increased but was still
below the production of orchards that had not incurred
vole damage. Total losses for the 2-year period were
estimated at $6,100 per acre ($15,250/ha) (Askham 1988).
Similar apple orchard loss figures were calculated for
pine voles in New York. Known densities of voles (0,
109, 218, and 436 per acre [0, 273, 545, and 1,090/ha])
were stocked in fenced blocks of McIntosh trees for 2
years. There was little impact the first year. The
second year, the highest vole population reduced fruit
yield 65.5% and increased undersized fruit from 3.1% to
57.5%. These factors caused a $2,745 per acre
($6,863/ha) reduction in income. In addition, survival
of the trees through a third year was considered
unlikely. The worst vole outbreak in the United States
probably occurred in Nevada in 1908 and 1909. Ten
thousand acres (400 ha) of alfalfa were completely
destroyed. Vole populations were estimated at 25,000 per
acre (62,500/ha).
Often a control program
may not appear to be justified in comparison to the
damage being incurred. It should be remembered, however,
that the “ounce of prevention” rule frequently applies
in vertebrate pest control. Preventive control measures
that at first appear too costly may eventually prove to
be a bargain.
Acknowledgments
I wish to thank Terry
Salmon, Bob Timm, Larry Blalock, and Robert Bechtel for
reviewing the first drafts of this chapter. Scott
Hygnstrom and anonymous reviewers improved the second
version of this chapter — thank you. Finally, I wish to
thank Miladene McCay and Linda Lesi for typing the first
and second versions.
Figures 1 and 5 from
Schwartz and Schwartz (1981).
Figures 2 through 4
adapted from Johnson and Johnson (1982) by Dave
Thornhill, University of Nebraska-Lincoln.
For Additional Information
Askham, L. E. 1988. A
two-year study of the physical and economic impact of
voles (Microtus montanus) on mixed maturity apple (Malus
spp.) orchards in the Pacific northwestern United
States. Proc. Vertebr. Pest. Conf. 13:151-155.
Burt, W. H., and R. P.
Grossenheider. 1976. A field guide to the mammals, 3d
ed. Houghton Mifflin Company, Boston. 189 pp.
Byers, R. E. 1985.
Management and control. Pages 621-646 in R. A. Tamarin,
ed. Biology of new world Microtus. Am. Soc. Mammal.
Shippensburg, Pennsylvania.
Byers, R. E., and M. H.
Merson 1982. Current improvements in baiting pine and
meadow voles. Proc. Vertebr. Pest Conf. 10:139-142.
Clark, J. P. 1986.
Vertebrate pest control handbook. California Dep. Food
Agric. Sacramento, California 610 pp.
Cole, R., and G. O. Batzli.
1979. Nutrition and population dynamics of the prairie
vole Microtus ochrogaster in central Illinois. Appl.
Ecol. 48:455-470.
Hall, E. R. 1981. The
mammals of North America, Vol. 2, 2d ed. John Wiley &
Sons, New York, Pp. 601-1181.
Jameson, E. W. 1947.
Natural history of the prairie vole. Univ. Kansas Publ.
Museum Nat. Hist. 1(7):125-151.
Johnson, E. W.. 1958.
Consumption of alfalfa and wild oats by Microtus
californicus. J. Wildl. Manag. 22:433-435.
Johnson, M. L., and S.
Johnson. 1982. Voles. Pages 326-354 in J. A. Chapman and
G. A. Feldhammer, eds. Wild mammals of North America:
biology, management and economics. The Johns Hopkins
Univ. Press, Baltimore, Maryland.
Libby, J. L., and J. I.
Abrams. 1966. Anticoagulant rodenticide in paper tubes
for control of meadow mice. J. Wildl. Manage.
30:512-518.
Marsh, R. E., R. E. Cole,
and W. E. Howard. 1967. Laboratory tests on the
effectiveness of prolin mouse tubes. J. Wildl. Manage.
31:342-344.
Pearce, J. 1947.
Identifying injury by wildlife to trees and shrubs in
northeast forests. US Dep. Inter. Fish Wildl. Serv.,
Washington, DC. Res. Rep. 13. 29 pp.
Pearson, O. P. 1985.
Predation. Pages 535-566 in R. A. Tamarin, ed. Biology
of new world Microtus. Amer. Soc. Mammal. Shippensburg,
Pennsylvania.
Radvanyi, A. 1980. Control
of small mammal damage in the Alberta oil sands
reclamation and afforestation program. For. Sci.
26:687-702.
Reich, L. M. 1981.
Microtus pennsylvanicus. Mammal. Species 159:1-8.
Richmond, M. E., C. G.
Forshey, L. A. Mahoffy, and P. N. Miller. 1987. Effects
of differential pine vole populations on growth and
yield of McIntosh apple trees. Proc. Eastern Wildl.
Damage Control Conf. 3:296-304.
Schwartz, C. W., and E. R.
Schwartz. 1981. The wild mammals of Missouri, rev. ed.
Univ. Missouri Press, Columbia. 356 pp.
Smolen, M. J. 1981.
Microtus pinetorum. Mammal. Species 147:1-7.
Smolen, M. J., and B. L.
Keller. 1987. Microtus longicaudus. Mammal. Species. 27
1:1-7.
Taitt, M. J., and C. J.
Krebs. 1985. Population dynamics and cycles. Pages
567-620 in R. A. Tamarin, ed., Biology of new world
Microtus. Amer. Soc. Mammal. Shippensburg, Pennsylvania.
White, L. 1965. Biological
and ecological considerations in meadow mouse population
management. Bull. California Dep. Agric. 54:161-171.
Editors
Scott E. Hygnstrom, Robert
M. Timm, Gary E. Larson
PREVENTION AND CONTROL OF
WILDLIFE DAMAGE — 1994
Cooperative Extension
Division Institute of Agriculture and Natural Resources
University of Nebraska -Lincoln
United States Department
of Agriculture Animal and Plant Health Inspection
Service Animal Damage Control
Great Plains Agricultural
Council Wildlife Committee
03/12/2007
Special
thanks to:
Clemson University
|